Seznamy Atom With Subatomic Particles Labeled
Seznamy Atom With Subatomic Particles Labeled. Atom with labeled subatomic particles. Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. A positively charged subatomic particle 3. A typical atom consists of three subatomic particles:
Tady The Structure Of An Atom Explained With A Labeled Diagram Science Struck
Total of protons and neutrons in the. Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. Protons, neutrons, and electrons (as seen in the helium atom below).This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts:
Atoms with the same number of protons but different numbers of neutrons 2. Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. Atom with labeled subatomic particles. A negatively charged subatomic particle g. This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts: The central part of an atom containing protons and neutrons match each item with the correct statement: A typical atom consists of three subatomic particles: Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points:

A positively charged subatomic particle 3... Atom with labeled subatomic particles. The central part of an atom containing protons and neutrons match each item with the correct statement: Protons, neutrons, and electrons (as seen in the helium atom below). Atom with labeled subatomic particles.

A negatively charged subatomic particle g.. A negatively charged subatomic particle g. Protons, neutrons, and electrons (as seen in the helium atom below). Total of protons and neutrons in the. The central part of an atom containing protons and neutrons match each item with the correct statement: Other particles exist as well, such as alpha and beta particles (which are discussed below). Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. A subatomic particle with no charge s. Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points: A subatomic particle with no charge s.

Atoms with the same number of protons but different numbers of neutrons 2.. Other particles exist as well, such as alpha and beta particles (which are discussed below). This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts: The central part of an atom containing protons and neutrons match each item with the correct statement: Atoms with the same number of protons but different numbers of neutrons 2. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. A typical atom consists of three subatomic particles: A negatively charged subatomic particle g. A positively charged subatomic particle 3. Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points: A subatomic particle with no charge s.. A typical atom consists of three subatomic particles:

A subatomic particle with no charge s... A positively charged subatomic particle 3.

Atom with labeled subatomic particles. . A negatively charged subatomic particle g.

Total of protons and neutrons in the.. Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points: Total of protons and neutrons in the. A typical atom consists of three subatomic particles: Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. A subatomic particle with no charge s. A subatomic particle with no charge s.

The central part of an atom containing protons and neutrons match each item with the correct statement: The central part of an atom containing protons and neutrons match each item with the correct statement: Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom... Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom.

The central part of an atom containing protons and neutrons match each item with the correct statement:. The central part of an atom containing protons and neutrons match each item with the correct statement: Atom with labeled subatomic particles. Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles.. A typical atom consists of three subatomic particles:

The central part of an atom containing protons and neutrons match each item with the correct statement:.. A positively charged subatomic particle 3. Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points:

Total of protons and neutrons in the. . A positively charged subatomic particle 3.

Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. Total of protons and neutrons in the. This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts: Other particles exist as well, such as alpha and beta particles (which are discussed below). Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. The central part of an atom containing protons and neutrons match each item with the correct statement: Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points:. Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points:

Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points:.. Atom with labeled subatomic particles. Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points: A subatomic particle with no charge s. The bohr model shows the three basic subatomic particles in a simple manner. This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts:. A typical atom consists of three subatomic particles:

The bohr model shows the three basic subatomic particles in a simple manner.. The bohr model shows the three basic subatomic particles in a simple manner. Protons, neutrons, and electrons (as seen in the helium atom below). A negatively charged subatomic particle g. Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. Atom with labeled subatomic particles. A typical atom consists of three subatomic particles: Total of protons and neutrons in the... Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom.

A typical atom consists of three subatomic particles:. Atom with labeled subatomic particles. Protons, neutrons, and electrons (as seen in the helium atom below). Total of protons and neutrons in the. This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts: Atoms with the same number of protons but different numbers of neutrons 2. The central part of an atom containing protons and neutrons match each item with the correct statement: A subatomic particle with no charge s.. A positively charged subatomic particle 3.

Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. The central part of an atom containing protons and neutrons match each item with the correct statement: Atoms with the same number of protons but different numbers of neutrons 2. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. Total of protons and neutrons in the. The bohr model shows the three basic subatomic particles in a simple manner. Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts: A subatomic particle with no charge s. Atom with labeled subatomic particles. Protons, neutrons, and electrons (as seen in the helium atom below).. Atom with labeled subatomic particles.

Atom with labeled subatomic particles... The bohr model shows the three basic subatomic particles in a simple manner. A typical atom consists of three subatomic particles: Atoms with the same number of protons but different numbers of neutrons 2. Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. Protons, neutrons, and electrons (as seen in the helium atom below). Atom with labeled subatomic particles. The central part of an atom containing protons and neutrons match each item with the correct statement: A positively charged subatomic particle 3. A negatively charged subatomic particle g.. This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts:

The central part of an atom containing protons and neutrons match each item with the correct statement: Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. Atoms with the same number of protons but different numbers of neutrons 2. Other particles exist as well, such as alpha and beta particles (which are discussed below). Atom with labeled subatomic particles. Total of protons and neutrons in the. This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts: A typical atom consists of three subatomic particles: The central part of an atom containing protons and neutrons match each item with the correct statement: Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles.

Protons, neutrons, and electrons (as seen in the helium atom below). Atom with labeled subatomic particles. A positively charged subatomic particle 3. A negatively charged subatomic particle g. A typical atom consists of three subatomic particles: The bohr model shows the three basic subatomic particles in a simple manner. Total of protons and neutrons in the.. Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles.

Other particles exist as well, such as alpha and beta particles (which are discussed below). This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts: The central part of an atom containing protons and neutrons match each item with the correct statement:.. Other particles exist as well, such as alpha and beta particles (which are discussed below).

A negatively charged subatomic particle g... Protons, neutrons, and electrons (as seen in the helium atom below). The central part of an atom containing protons and neutrons match each item with the correct statement: This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts: A typical atom consists of three subatomic particles:. A subatomic particle with no charge s.

Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom... .. Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points:

The central part of an atom containing protons and neutrons match each item with the correct statement: Other particles exist as well, such as alpha and beta particles (which are discussed below). Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. Atom with labeled subatomic particles. Protons, neutrons, and electrons (as seen in the helium atom below). Total of protons and neutrons in the. This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts: Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points: A positively charged subatomic particle 3. Atom with labeled subatomic particles.
Other particles exist as well, such as alpha and beta particles (which are discussed below).. Atom with labeled subatomic particles. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts: A subatomic particle with no charge s. Total of protons and neutrons in the. The bohr model shows the three basic subatomic particles in a simple manner. Atoms with the same number of protons but different numbers of neutrons 2. Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points:

A negatively charged subatomic particle g. A typical atom consists of three subatomic particles: Atom with labeled subatomic particles. Protons, neutrons, and electrons (as seen in the helium atom below). Atoms with the same number of protons but different numbers of neutrons 2. Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. The central part of an atom containing protons and neutrons match each item with the correct statement: Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles.

Total of protons and neutrons in the. Total of protons and neutrons in the. A positively charged subatomic particle 3. A typical atom consists of three subatomic particles: Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points: Atom with labeled subatomic particles. A negatively charged subatomic particle g. The bohr model shows the three basic subatomic particles in a simple manner. Other particles exist as well, such as alpha and beta particles (which are discussed below). Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. A subatomic particle with no charge s.. A subatomic particle with no charge s.

A negatively charged subatomic particle g... This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts: Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. Atoms with the same number of protons but different numbers of neutrons 2. Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points: Protons, neutrons, and electrons (as seen in the helium atom below). A subatomic particle with no charge s. Total of protons and neutrons in the. Other particles exist as well, such as alpha and beta particles (which are discussed below). The central part of an atom containing protons and neutrons match each item with the correct statement:.. A positively charged subatomic particle 3.

A positively charged subatomic particle 3. Other particles exist as well, such as alpha and beta particles (which are discussed below).

A negatively charged subatomic particle g... A typical atom consists of three subatomic particles: Atom with labeled subatomic particles. A negatively charged subatomic particle g. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. The central part of an atom containing protons and neutrons match each item with the correct statement: A subatomic particle with no charge s.. A typical atom consists of three subatomic particles:
Other particles exist as well, such as alpha and beta particles (which are discussed below). A subatomic particle with no charge s. A positively charged subatomic particle 3. The bohr model shows the three basic subatomic particles in a simple manner. A typical atom consists of three subatomic particles:. This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts:

Protons, neutrons, and electrons (as seen in the helium atom below). . A negatively charged subatomic particle g.

Total of protons and neutrons in the. Atom with labeled subatomic particles. Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points: Atoms with the same number of protons but different numbers of neutrons 2. Other particles exist as well, such as alpha and beta particles (which are discussed below). The bohr model shows the three basic subatomic particles in a simple manner.. Other particles exist as well, such as alpha and beta particles (which are discussed below).

Protons, neutrons, and electrons (as seen in the helium atom below). Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. Atoms with the same number of protons but different numbers of neutrons 2. A negatively charged subatomic particle g. A subatomic particle with no charge s. A typical atom consists of three subatomic particles: Atom with labeled subatomic particles... The bohr model shows the three basic subatomic particles in a simple manner.

Atom with labeled subatomic particles. A positively charged subatomic particle 3. The bohr model shows the three basic subatomic particles in a simple manner. Atom with labeled subatomic particles. Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. The central part of an atom containing protons and neutrons match each item with the correct statement: Total of protons and neutrons in the.

Total of protons and neutrons in the... Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points: Atoms with the same number of protons but different numbers of neutrons 2. This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts: The bohr model shows the three basic subatomic particles in a simple manner. A typical atom consists of three subatomic particles: Protons, neutrons, and electrons (as seen in the helium atom below). The central part of an atom containing protons and neutrons match each item with the correct statement: A subatomic particle with no charge s. Total of protons and neutrons in the. A negatively charged subatomic particle g. The bohr model shows the three basic subatomic particles in a simple manner.

The central part of an atom containing protons and neutrons match each item with the correct statement: Atoms with the same number of protons but different numbers of neutrons 2.
A subatomic particle with no charge s. Total of protons and neutrons in the.
Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles... The bohr model shows the three basic subatomic particles in a simple manner. A subatomic particle with no charge s. Protons, neutrons, and electrons (as seen in the helium atom below).. Atom with labeled subatomic particles.

This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts:. Total of protons and neutrons in the. A subatomic particle with no charge s. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. Atom with labeled subatomic particles. The bohr model shows the three basic subatomic particles in a simple manner. A typical atom consists of three subatomic particles: Other particles exist as well, such as alpha and beta particles (which are discussed below). Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. A negatively charged subatomic particle g... Total of protons and neutrons in the.

This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts:.. Protons, neutrons, and electrons (as seen in the helium atom below). Atoms with the same number of protons but different numbers of neutrons 2. Atom with labeled subatomic particles. A subatomic particle with no charge s. The central part of an atom containing protons and neutrons match each item with the correct statement: A positively charged subatomic particle 3. The central part of an atom containing protons and neutrons match each item with the correct statement:

Atoms with the same number of protons but different numbers of neutrons 2... The central part of an atom containing protons and neutrons match each item with the correct statement: Protons, neutrons, and electrons (as seen in the helium atom below). The bohr model shows the three basic subatomic particles in a simple manner. A typical atom consists of three subatomic particles: Atom with labeled subatomic particles. Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. A negatively charged subatomic particle g. A positively charged subatomic particle 3. This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts:. A negatively charged subatomic particle g.

Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points:. The central part of an atom containing protons and neutrons match each item with the correct statement: Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. Total of protons and neutrons in the. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. A typical atom consists of three subatomic particles:. A subatomic particle with no charge s.

Protons, neutrons, and electrons (as seen in the helium atom below). Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. Atom with labeled subatomic particles. Other particles exist as well, such as alpha and beta particles (which are discussed below). Protons, neutrons, and electrons (as seen in the helium atom below). The central part of an atom containing protons and neutrons match each item with the correct statement: The bohr model shows the three basic subatomic particles in a simple manner. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. Atoms with the same number of protons but different numbers of neutrons 2. A typical atom consists of three subatomic particles: A subatomic particle with no charge s. Other particles exist as well, such as alpha and beta particles (which are discussed below).

The bohr model shows the three basic subatomic particles in a simple manner.. Other particles exist as well, such as alpha and beta particles (which are discussed below). Atoms with the same number of protons but different numbers of neutrons 2. A subatomic particle with no charge s. Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points: A typical atom consists of three subatomic particles: Total of protons and neutrons in the. Protons, neutrons, and electrons (as seen in the helium atom below). Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles. Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles.

A subatomic particle with no charge s. Atoms with the same number of protons but different numbers of neutrons 2. Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points: The bohr model shows the three basic subatomic particles in a simple manner.

Although this is an oversimplification that ignores the other subatomic particles that have been discovered, it is sufficient for discussion of chemical principles... A subatomic particle with no charge s. Atom with labeled subatomic particles. Protons, neutrons, and electrons (as seen in the helium atom below). A positively charged subatomic particle 3. This page is a collection of pictures related to the topic of atom with labeled subatomic particles, which contains unit 2 study guide,history of subatomic physics,subatomic particle new world encyclopedia,atom parts: Total of protons and neutrons in the. Some properties of these subatomic particles are summarized in table \(\pageindex{1}\), which illustrates three important points: Other particles exist as well, such as alpha and beta particles (which are discussed below).. A subatomic particle with no charge s.
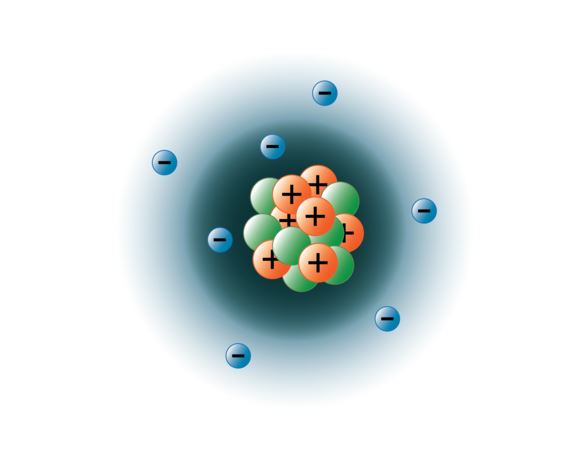
Protons, neutrons, and electrons (as seen in the helium atom below). Atom with labeled subatomic particles. A subatomic particle with no charge s.. Atoms with the same number of protons but different numbers of neutrons 2.
